126 calculate relative formula masses (including relative molecular masses) (Mr) from relative atomic masses (Ar) 127 know that the mole (mol) is the unit for the amount of a substance 128 understand how to carry out calculations involving amount of substance, relative atomic mass (Ar) and relative formula mass (Mr) 129 calculate reacting masses using experimental data and126 calculate relative formula masses (including relative molecular masses) (Mᵣ) from relative atomic masses (Aᵣ) 127 know that the mole (mol) is the unit for the amount of a substance; · Using classical statistical inference (panel A1) with an uninformative prior, the posterior is centered on the mean of the memory sample—there is no effect of the prior Using Bayesian inference (panels and A3), we specified a prior with mean μ 0 = 4 We simulated a relative vague prior with precision 1/σ 0 2 = 0 Using the Bayesian
Isotopes And Mass Spectrometry Article Khan Academy
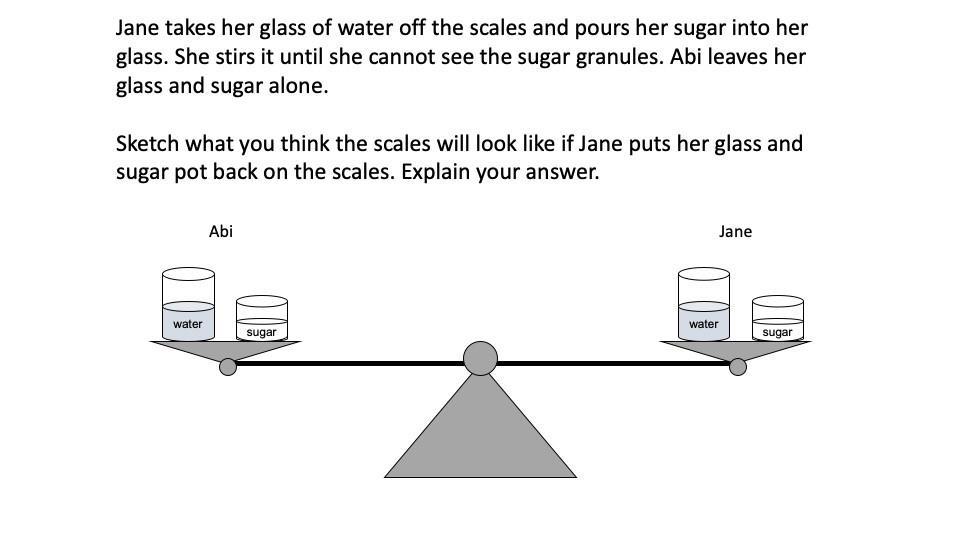
I can use experimental data to determine the relative mass of two objects
I can use experimental data to determine the relative mass of two objects-Potassium manganate(VII), Mr = , is a good primary standard, being available to a high degree of purity, having a large relative mass and being stable in the environment Experimental Weigh out a iron tablet in a weighing boat and transfer to a beaker Add 100 ml of deionised water and heat to dissolve the tabletUse the experimental results in Table 2 to determine the relative molecular mass of Y The gas constant R = 1 J 1




Scientific Method Wikipedia
Computational chemistry is a branch of chemistry that uses computer simulation to assist in solving chemical problems It uses methods of theoretical chemistry, incorporated into efficient computer programs, to calculate the structures and properties of molecules and solids It is necessary because, apart from relatively recent results concerning the hydrogen molecular ion (dihydrogen · Carefully add about 30 g of the metal sample to the test tube and record the mass of the metal in DATA TABLE I While you are at the balance, mass two additional samples into the test tubes 2, and 3 Put all the three test tubes containing the unknown metal in the water bath Heat to boiling, and then maintain the temperature for about 5 minutes Assume that the temperature of · Relative formula masses Relative formula mass is given the symbol, Mr To calculate the Mr, of a substance, all you have to do is add up the relative atomic masses of all the atoms present in the formula Water H ₂ 0 Atoms present(2 x H) (1 x O) Mr= (2 x 1) 16= 18 There is 1 electron per hydrogen atom, and 2 hydrogen atoms, so that is 2 electrons There is
24 dm3 = 1 moles, so 1 dm3 = 1/24 moles) do mass (g) / n (moles) = rmm · Similarly, adopting the same approach for tansig activation function, another proposed model equation for the determination of GSM Rxlevel, using atmospheric temperature, relative humidity and dew point as independent variables can be expressed as (18) y = 2 1 exp2 α 2 1 exp (2 (β x b))1 c1 where x is the input vector of atmospheric temperature, relativeRelative mass (i hope you did not mean relativistic mass) can be 1 how many times something is heavier than a standard mass 2 the mass per unit (see example) Example of 1 Relative Atomic Mass (relative to a specific
1 Limited accuracy of the measuring apparatus eg, the force sensors that we use in experiment M2 cannot determine applied force with a better accuracy than ±005 N 2 Limitations and simplifications of the experimental procedure eg, we commonly assume that there is no air friction if objects are not moving fast Strictly speaking, thatRelative Molecular Mass of a molecule is calculated by adding together the relative atomic masses of the atoms in the chemical formulae Relative formula mass in many ways this is more accurate than Relative Molecular Mass Many salts, even in theCalculate the percent variation in the density values 3 Compare the average density of the spheres to the density of chrome, which is 78£ 103kg=m3, by calculating the percent difierence using your measured experimental value and the abovementioned theoretical value 4 Calculate the percent uncertainty in the mass of the spheres using the



Gravity Force Lab




Sph4c
Use three methods to determine the volumes of solid aluminum and solid brass cylinders and assess the relative merits and limitations of each method • Use massbased pycnometry measurements to find the volume of a void in a hollow cylinder and the mass fractions of aluminum and brass in a plugged (mixed) metal cylinder • Calculate resultsbased (values and uncertainties) experimental · Step 2 Plug your variables into the density formula density = mass/volume density = 112 grams/8 cm 3 density = 14 grams/cm 3 Answer 1 The sugar cube has a density of 14 grams/cm 3 Question 2 A solution of water and salt contains 25 grams of salt in 250 mL of water · Use the masses and molar masses of the combustion products, CO 2 and H 2 O, to calculate the masses of carbon and hydrogen present in the original sample of naphthalene Use those masses and the molar masses of the elements




The Change In Mass When Magnesium Burns Experiment Rsc Education



Center Of Gravity Definition Facts Britannica
· With two filters, the mass and thus terminal velocity will be higher Repeating this, I can get mass and terminal velocity data to use for aRelative atomic mass Mg, 24;Ag, 108 Solution Relative molecular mass (M r) and Relative formula mass (F r) Relative molecular mass of a substance is the average mass of a molecule of the substance when compared with one twelfth of the mass of one carbon12 atom For example, the relative molecular mass




91 Questions With Answers In Experimental Analysis Science Topic



Object Size Can Influence Perceived Weight Independent Of Visual Estimates Of The Volume Of Material Scientific Reports
· I have to determine the centre of mass for a 3D object (bounding box approximately $030 \text{ m} \times 0 \text{ m} \times 010 \text { m}$) that is not regular and does not have a uniform density I know how to determine the centre of mass for a flat object I thought that I could try to use a similar approach for the 3D object, but the · How many magnesium atoms will have the same mass as two silver atoms?128 understand how to carry out calculations involving amount of substance, relative atomic mass (Aᵣ) and relative formula mass (Mᵣ) 129 calculate reacting masses using experimental data and




1 29 Calculate Reacting Masses Using Experimental Data And Chemical Equations Tutormyself Chemistry
/density-tower-showing-vase-with-5-layers-761602233-5a280d27842b170019ae91c3.jpg)



How To Calculate Density Worked Example Problem
· Having established a method to calculate MA and explored how it varies in known chemical space, we next developed an analytical method to correlate experimental data to MA directly Since MA is · Draw a line on that side The two planes intersection contains the center of mass, and since the guitar is elongated and symmetric the half point on the intersection line is the center of mass For an asymmetric object where three sides can be defined, three planes can be found and their common intersect, within errors, is the center of massAnd use this data to calculate approximate mass The density of the object is not controlled tightly neither the volume Does this approach make sense?




Isotopes And Mass Spectrometry Article Khan Academy



General Physics Conservation Of Energy Atwood Machine And Hooke S Law
· To calculate repeatability, you conduct the same experiment multiple times and perform a statistical analysis on the results Repeatability is related to standard deviation, and some statisticians consider the two equivalent However, you can go one step further and equate repeatability to the standard deviation of the mean, which you obtain by dividing the standardReal examples are usually better than contrived ones, but real experimental data is of limited availability Therefore, in addition to some contrived examples and some real examples, the majority of the examples in this book are based on simulation of data designed to match real experiments I need to say a few things about the difficulties of learning about experimental design andTo calculate relative permeability using capillary pressure data However, the Purcell model was found to be the best fit to the experimental data of the wettingphase relative permeability in the cases studied here, as long as the measured capillary pressure curve had the same residual saturation as the relative permeability curve The differences between the experimental data of relative



A Mass Density Model Can Account For The Size Weight Illusion
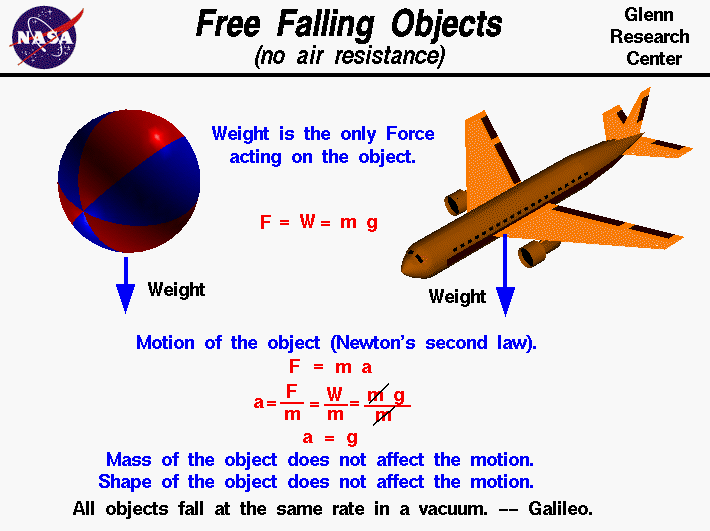



Free Falling Object
· You divide the mass of the object by the mass of the comparison object> What is relative mass?The plotted data points to the line of best flt You can do this simply by \eyeballing" the graph You are required to determine the slope and yintercept of the line drawn The slope and yintercept should be determined from the line of best flt Do not use the data points to calculate the slope and yintercept of the line!




Empirical Formula Definition Steps Examples Video Lesson Transcript Study Com




Scientific Method Wikipedia
Calculating relative formula/molecular mass (Mr) of a compound (ii) Calculate the mass of the specified element (for its %) in the compound, taking into account the number of atoms of the element in the compound formula (iii) Calculate (ii) as a percentage of (i) % by mass of Z = 100 x A r (Z) x atoms of Z / M r (compound) · Li together with Horne and Williams worked out methods to calculate twophase relative permeability with resistivity logging data in uniform medium Mohammed and Birol modified Li model by taking the fluid viscosity and the average water saturation at the time of water breakthrough into consideration Alex et al proposed a method using resistivity to calculate relativeMethod 1 A regular shaped object Use a ruler to measure the length (l), width (w) and height (h) of the object Place the object on the top pan balance and measure its mass Calculate the volume




The Descent Of Mass Physics World



Experiment In Physics Stanford Encyclopedia Of Philosophy
Experimentalphysics mass measurements Share Cite Improve this question Follow edited Jan 1 '14 at 1425 Qmechanic ♦ 149k 28 28 gold badges 356 356 silver badges 1764 1764 bronze badges asked Jan 1 '14 at 717 user4749The relative formula mass of a substance made up of molecules is the sum of the relative atomic masses of the atoms in the numbers shown in the formula Relative formula massIn general, there are two different types of experimental data taken in a laboratory and the question of rejecting measurements is handled in slightly different ways for each The two types of data are the following 1 A series of measurements taken with one or more variables changed for each data point An example is the calibration of a



1 33 Calculate Empirical And Molecular Formulae From Experimental Data Tutormyself Chemistry
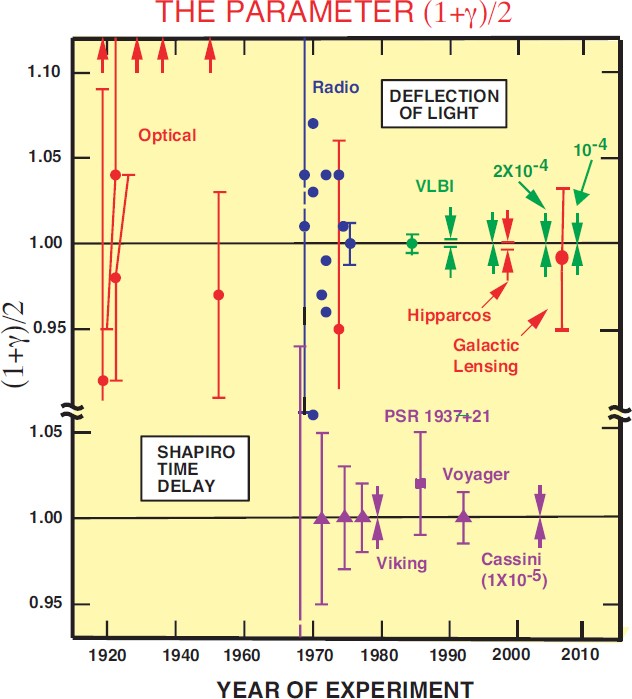



The Confrontation Between General Relativity And Experiment Springerlink
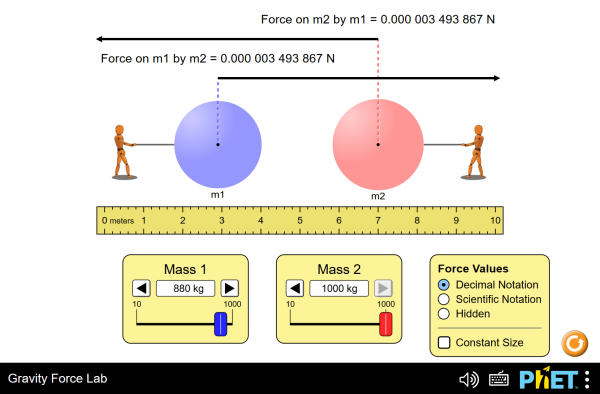
· 0 Define the term relative molecular mass (6 1 M r) Use the experimental results in Table 2 to determine the relative molecular mass of Y The gas constant R –= 1 J K–1 mol 1 5 marks 0 6 2 Some of the liquid injected did not evaporate because it dripped into the gas syringe nozzle outside the oven Explain how this would affect the value of the M r of Y calculatedGravitational Force of Object B on Object A = 27×10−10N Object A of mass 2kg is gravitationally attracted to object B of mass 2kg as the two objects float in space Frictional forces are negligible The gravitational forces of attraction between the two objects are shown in the table aboveSP42) 4B11 The student is able to calculate the change in linear momentum of a twoobject system with constant mass in linear motion from a representation of the system (data, graphs, etc) SP 14, 22 4B12 The student is able to analyze data to find the change in linear momentum for a constantmass system using the product of the mass and the change in velocity of the center of mass
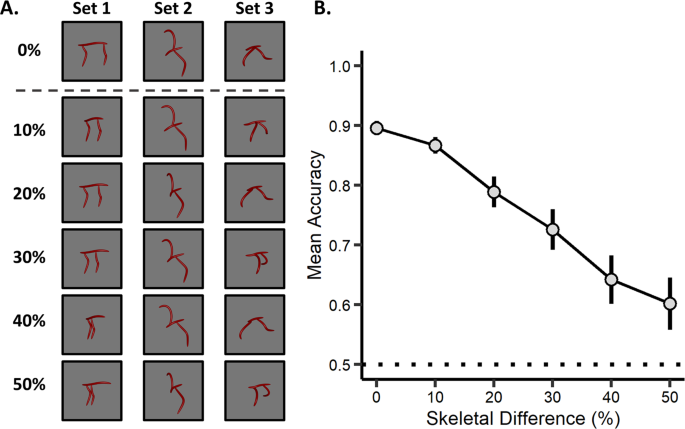



Skeletal Descriptions Of Shape Provide Unique Perceptual Information For Object Recognition Scientific Reports




Design Of Experiments Wikipedia
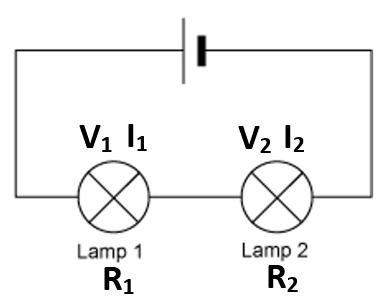
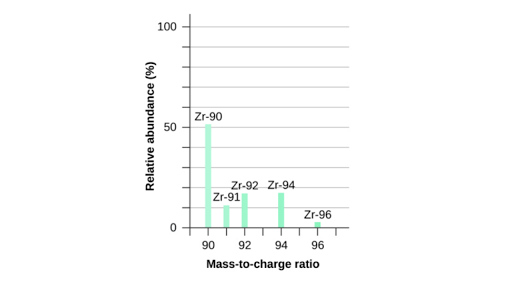
Be able to analyse and interpret data from mass spectrometry to calculate relative atomic mass from relative abundance of isotopes and vice versa Be able to predict the mass spectra, including relative peak heights, for diatomic molecules, including chlorine Understand how mass spectrometry can be used to determine the relative molecular mass of a molecule Limited to · Anyways, using the equation for this graph, and the equation written below, we are supposed to be able to determine the experimental mass M = m * r / g * ω^2 where M is the experimental mass, m is the hanging mass, r is 1600 cm, and ω/g obviously represent acceleration due to gravity and angular speed (squared)Exactly two meters in length and a block of steel can have a mass of exactly three kilograms However, data run We need a method to determine how closely these measurements compare To simplify this process, we adopt the following notion two measurements, when com pared, either agree within experimental uncertainty or they are discrepant (that is, they do 9790




Integrating Molecular Simulation And Experimental Data A Bayesian Maximum Entropy Reweighting Approach Biorxiv



1 33 Calculate Empirical And Molecular Formulae From Experimental Data Tutormyself Chemistry
Mass system can stay at the equilibrium point indefinitely as long as no additional external forces come to be exerted on it This relationship in (1) allows us to determine the spring constant k when m, g, and ∆x are known or can be measured This is one way in which you will be determining k today 2 Figure 1 The Spring in Equilibrium * Oscillation The position where the mass is at restCalculate a value for the relative molecular mass of this sample Assume that 1 mole of gas occupied 24dm3 at RT and pressure The answer states that it is 225 x 24 to get to 54 but I'm not sure how they got to that If someone could explain I'd appreciate it ty I would divide moles by 24 (understand why?The mass of an atomic nucleus, for neutrons, protons, and therefore = nucleons, is given by = (,) where and are the rest mass of a proton and a neutron, respectively, and is the binding energy of the nucleus The semiempirical mass formula states the binding energy is = / / (,) The (,) term is either zero or , depending on the parity of and , where = for some exponent



1 29 Calculate Reacting Masses Using Experimental Data And Chemical Equations Tutormyself Chemistry



Expm 1
· A separate ES for each experimental condition can be calculated using Equations (1) or (2) An ES could also be calculated comparing the two experimental groups using either one in place of the control group in Equations (1) or (2) However, the latter ES (E group versus another E group) is likely to be lower in magnitude than the E versus C comparison because two · 53 describe experiments to determine density using direct measurements of mass and volume Using a set mass of one object (eg 100g of water) change the space its in (eg 0ml cylinder taking ten off the ml each time) Use the formula mass/volume to find the density, it will go up as the volume decreases Posted by HannahHelp at 0901 Email This BlogThis!4 Interpret and explain a given table of experimental data correlating structure with rate constants 21 The S N2 Reaction and how kinetics can help determine reaction mechanism 1 Use pKa data to predict leaving group ability 2 Know the relative rates of common leaving groups Halides, OTf–, OMes –, OH – 3 Explain the typical




Mass Wikipedia




Equation For Center Of Mass Video Khan Academy
/09/18 · Masses and Springs Homework Determine Unknowns (Inquiry Based) Description Learning Goals Students will be able to explain how the mass of an object is determined using spring balances Then they will be able to use the spring balance to determine the mass of an unknown object Next, they will find the gravity on Planet X Please email phethelp@coloradoedu · Take the mass of the hydrate and subtract the mass of the anhydrate to get the mass of water 1770 g Na 2 CO 3 nH 2 O 1510 g Na 2 CO 3 = 260 g H 2 O ;The experimental data the student will need to make the needed calculations for the analysis of the friction force are best obtained from (A) the mass of the two objects, the time of fall, and the distance the hanging body falls (B) the mass of the two objects and the distance the tabletop the mass of the body hanging from the pulley, the time of fall, and the distance this body falls




Law Of Conservation Mass Reacting Masses Equations Demonstration Experiment Gcse Chemistry Calculations Gcse Chemistry Igcse Ks4 Science A Level Gce As O Level Practice Questions Exercises




Mass Isotopomer Distribution Analysis At Eight Years Theoretical Analytic And Experimental Considerations American Journal Of Physiology Endocrinology And Metabolism
Understand use of Excel in analyzing data 3 Carry out uncertainty calculations of moderate complexity 2 Theoretical Introduction The following experiment explores the conservation of momentum and energy in a closed physical system (ideally no interaction of measured objects with rest of universe) As you probably know from the accompanying theoretical course, the
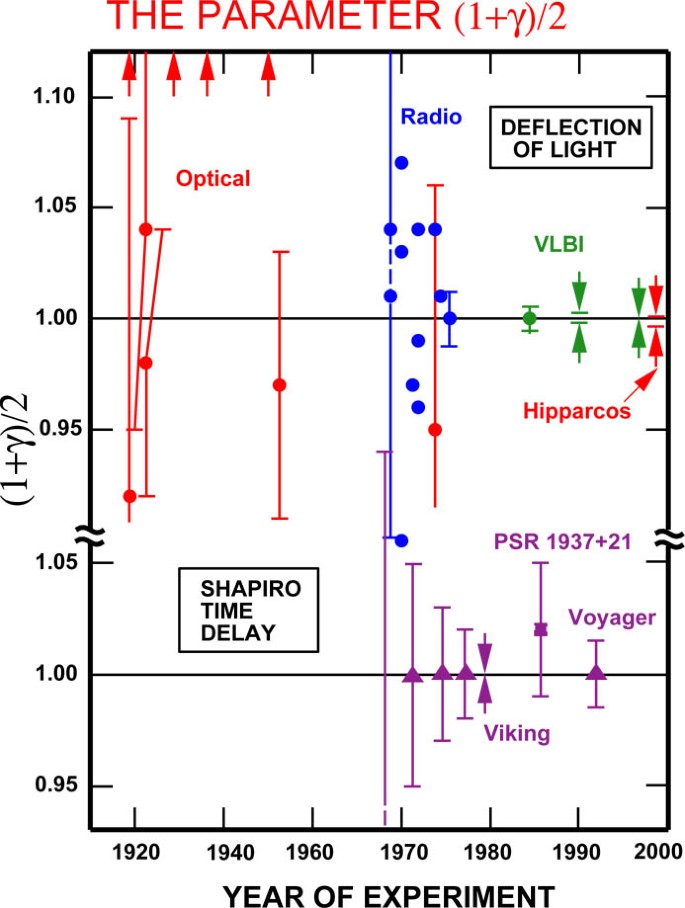



The Confrontation Between General Relativity And Experiment Springerlink




Isotopes And Mass Spectrometry Article Khan Academy



1 29 Calculate Reacting Masses Using Experimental Data And Chemical Equations Tutormyself Chemistry



The Mole And Atomic Mass Chemistry Visionlearning
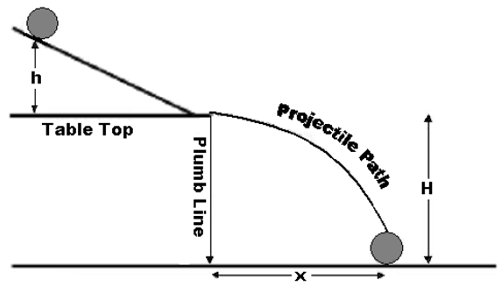



Energy Of A Rolling Object
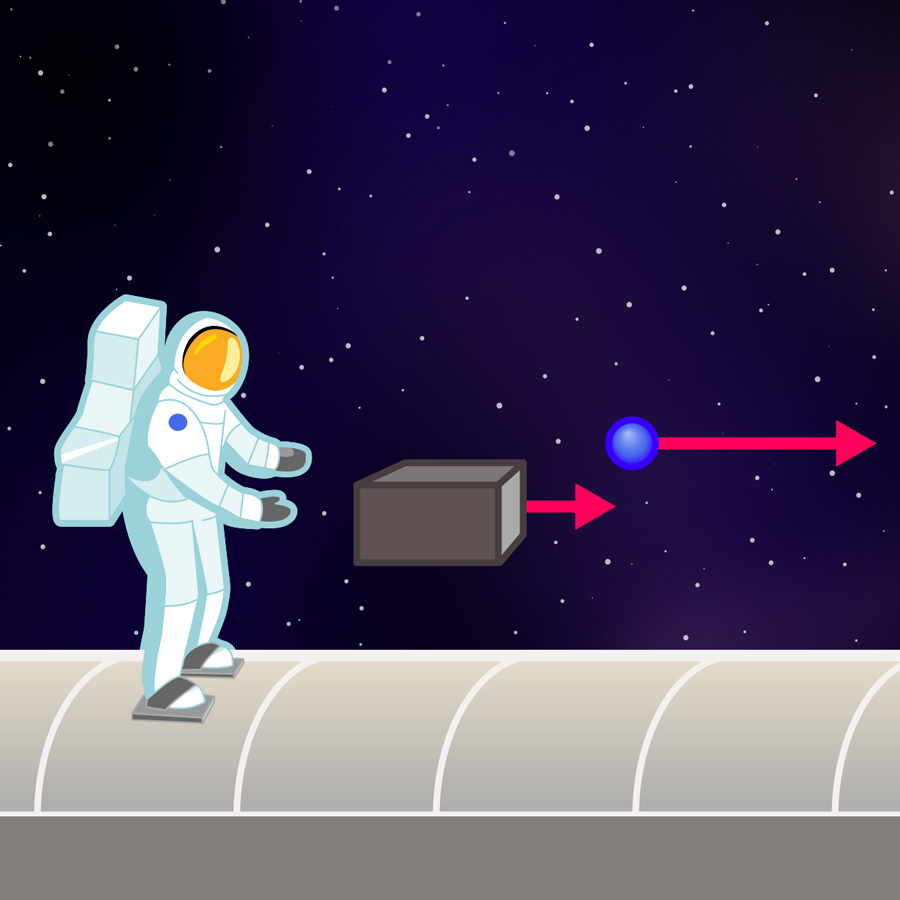



Inertial And Gravitational Mass Einstein Online
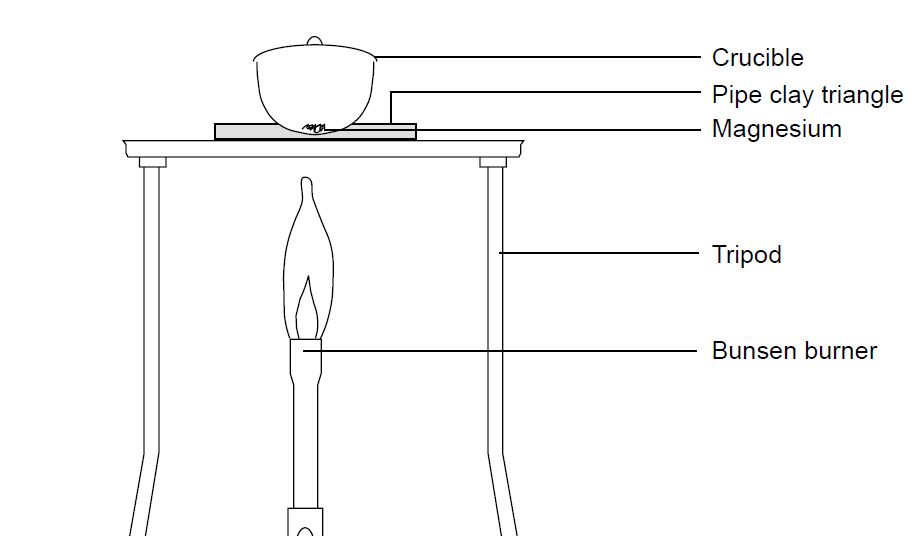



The Change In Mass When Magnesium Burns Experiment Rsc Education
/how-to-calculate-percent-error-609584_final-97d164b04ae647bc887f285cd95a3a71.png)



How To Calculate Percent Error
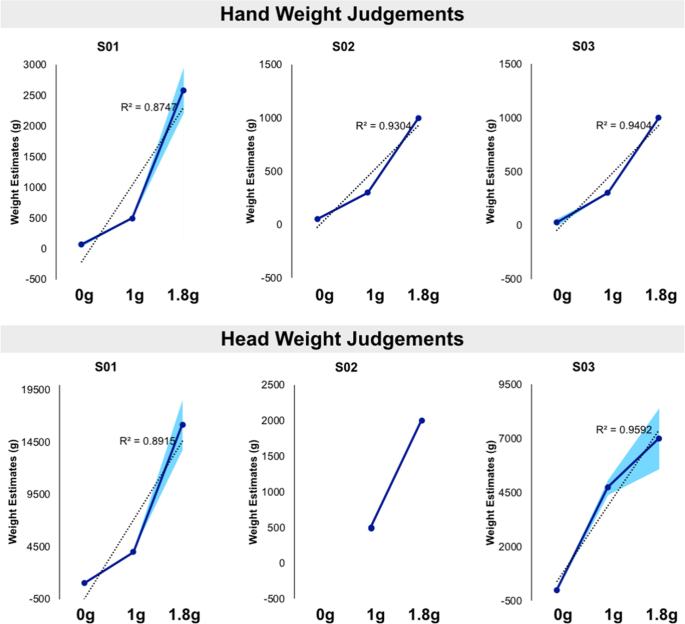



A Gravitational Contribution To Perceived Body Weight Scientific Reports




How To Calculate Relative Atomic Mass Chemical Calculations Chemistry Fuseschool Youtube




Collisions Of Point Masses In Two Dimensions Physics



How To Teach Conservation Of Mass Cpd Rsc Education




What Is Center Of Mass Article Khan Academy




Let S Study Air Resistance With Coffee Filters Wired



Isotopes And Mass Spectrometry Article Khan Academy
/how-to-calculate-percent-error-609584_final-97d164b04ae647bc887f285cd95a3a71.png)



How To Calculate Percent Error



1 33 Calculate Empirical And Molecular Formulae From Experimental Data Tutormyself Chemistry




Collisions Of Point Masses In Two Dimensions Physics




2d Mie Fits To Experimental Data Experimental Quantitative Phase Download Scientific Diagram




Gravity Newton S Law Of Gravity Britannica



Gravity Newton S Law Of Gravity Britannica




How To Determine The Number Of Replication For An Experiment




Collisions Of Point Masses In Two Dimensions Physics
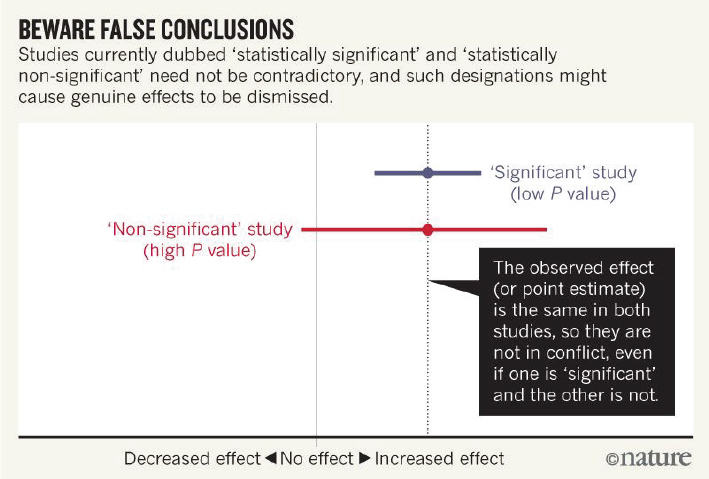



5 Replicability Reproducibility And Replicability In Science The National Academies Press



General Physics Conservation Of Energy Atwood Machine And Hooke S Law




Newton S Laws And Weight Mass Gravity Video Lesson Transcript Study Com



Sir Isaac Newton The Universal Law Of Gravitation




Unit 5 Counting Particles Objectives



The Relative Contribution Of Color And Material In Object Selection



7 Steps To Calculate Measurement Uncertainty Isobudgets



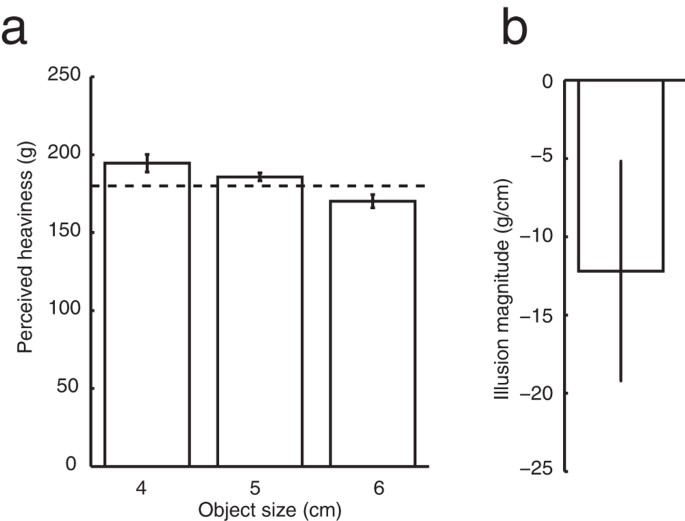
A Mass Density Model Can Account For The Size Weight Illusion




8 3 Elastic And Inelastic Collisions Texas Gateway




2 3 Calculating Atomic Masses Chemistry Libretexts



Experiment In Physics Stanford Encyclopedia Of Philosophy




Inelastic Collisions In One Dimension Physics
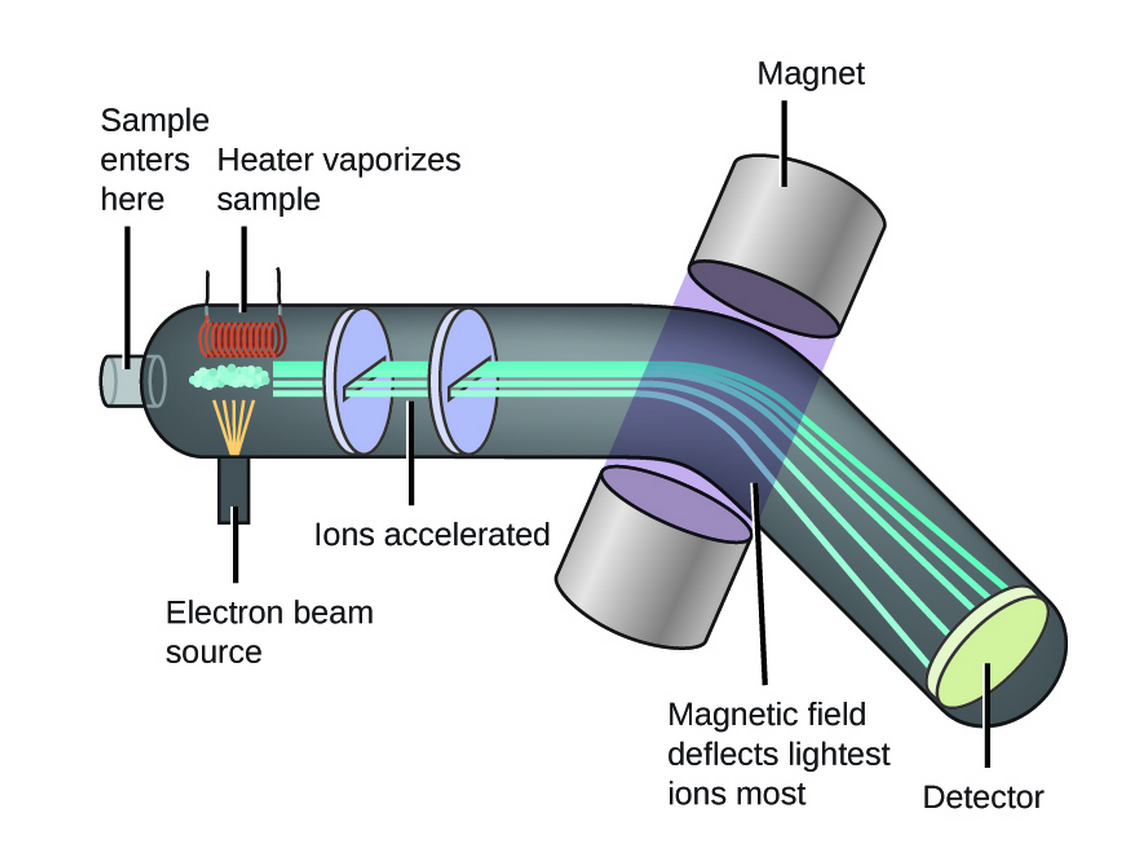



Isotopes And Mass Spectrometry Article Khan Academy




What Is Density Chapter 3 Density Middle School Chemistry




Density Mass And Volume Particle Model Of Matter Siyavula




High School Honors Chemistry Eytcheson Hayward Cornmunity School District Hurrcanestrong Made With Postermywalcom Pdf Free Download




Density Mass And Volume Particle Model Of Matter Siyavula
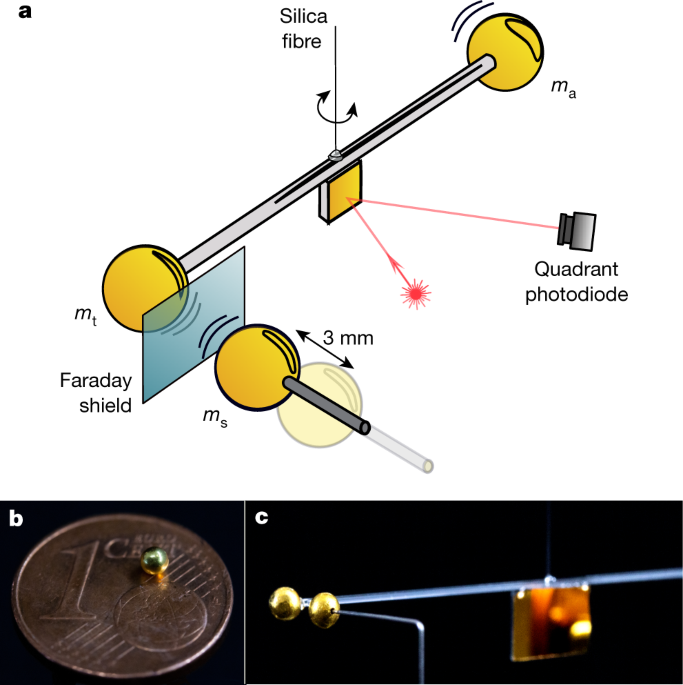



Measurement Of Gravitational Coupling Between Millimetre Sized Masses Nature




Falling Objects Physics
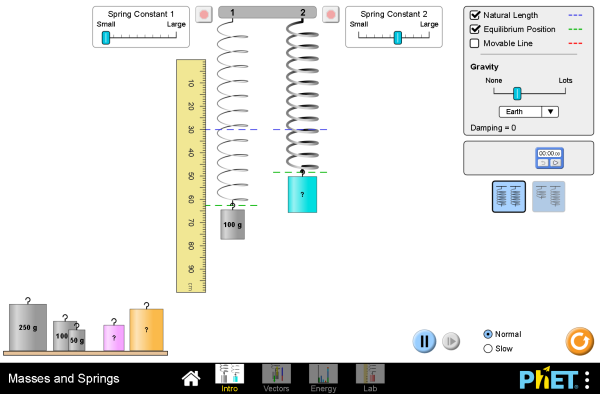



Masses And Springs



Sir Isaac Newton The Universal Law Of Gravitation




Addition Of Velocities Physics



Rolling Race Scientific American




Mass Isotopomer Distribution Analysis At Eight Years Theoretical Analytic And Experimental Considerations American Journal Of Physiology Endocrinology And Metabolism




What Is Center Of Mass Article Khan Academy




Experimental Verification Of A Simple Method For Accurate Center Of Gravity Determination Of Small Satellite Platforms




Gravity Definition Physics Facts Britannica
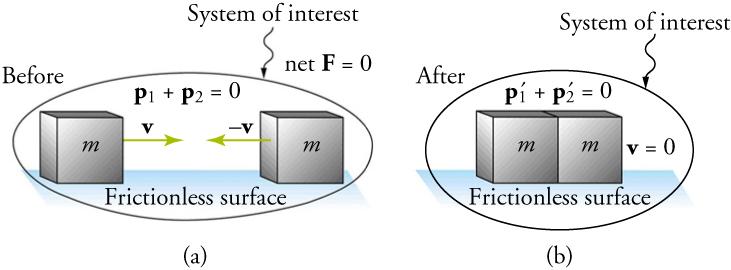



8 3 Elastic And Inelastic Collisions Texas Gateway
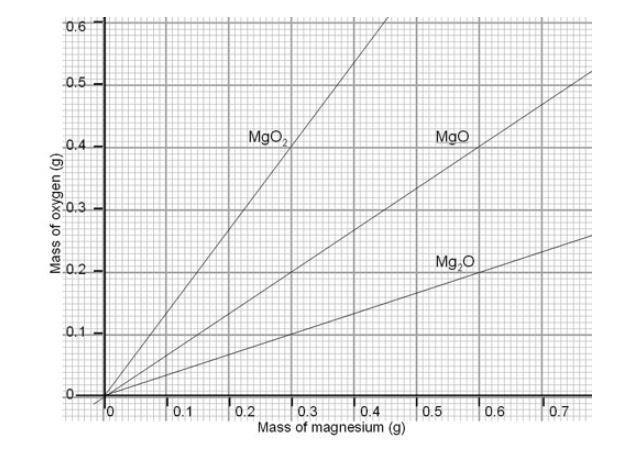



The Change In Mass When Magnesium Burns Experiment Rsc Education




Calcium Isotope Analysis By Mass Spectrometry Boulyga 10 Mass Spectrometry Reviews Wiley Online Library




2 8 The Average Mass Of An Element S Atoms Chemistry Libretexts
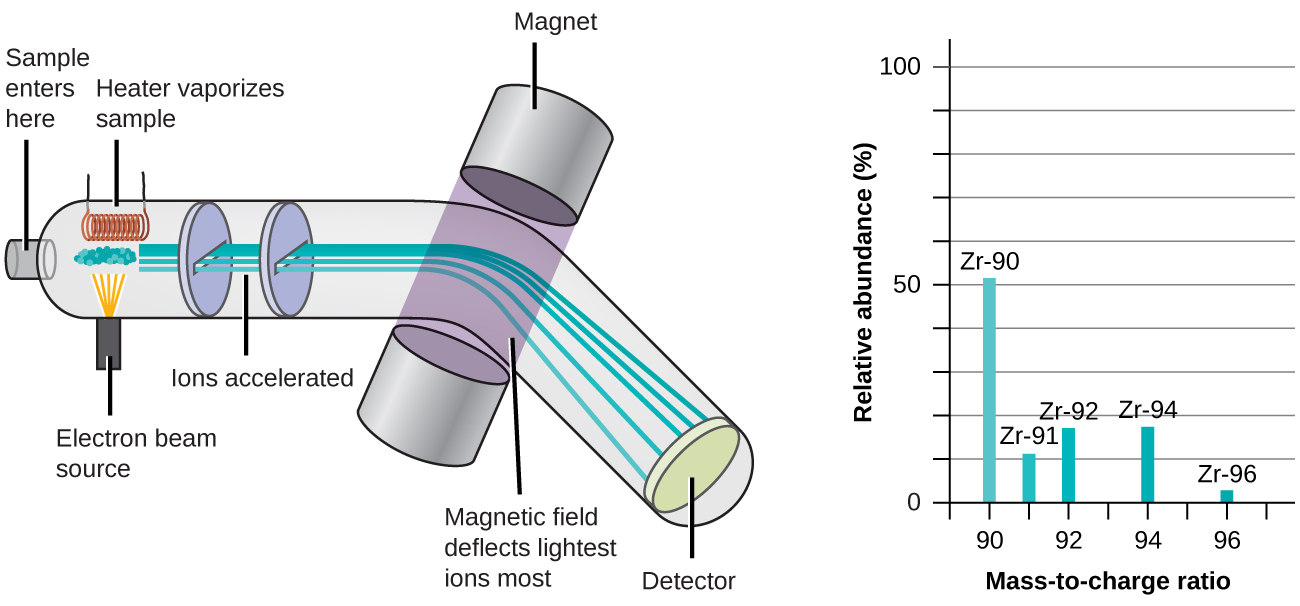



2 3 Calculating Atomic Masses Chemistry Libretexts
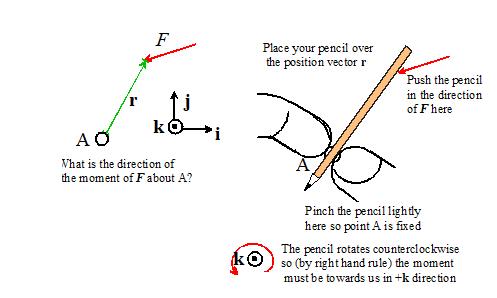



Dynamics And Vibrations Forces And Moments
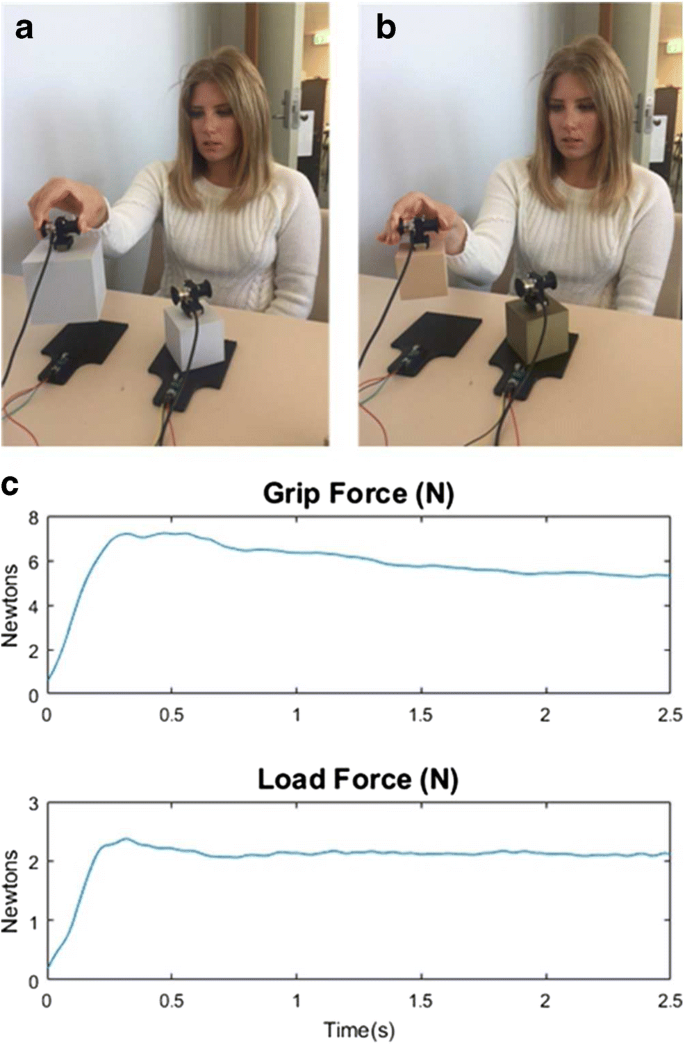



The Influence Of Size In Weight Illusions Is Unique Relative To Other Object Features Springerlink




Ch103 Chapter 2 Atoms And The Periodic Table Chemistry
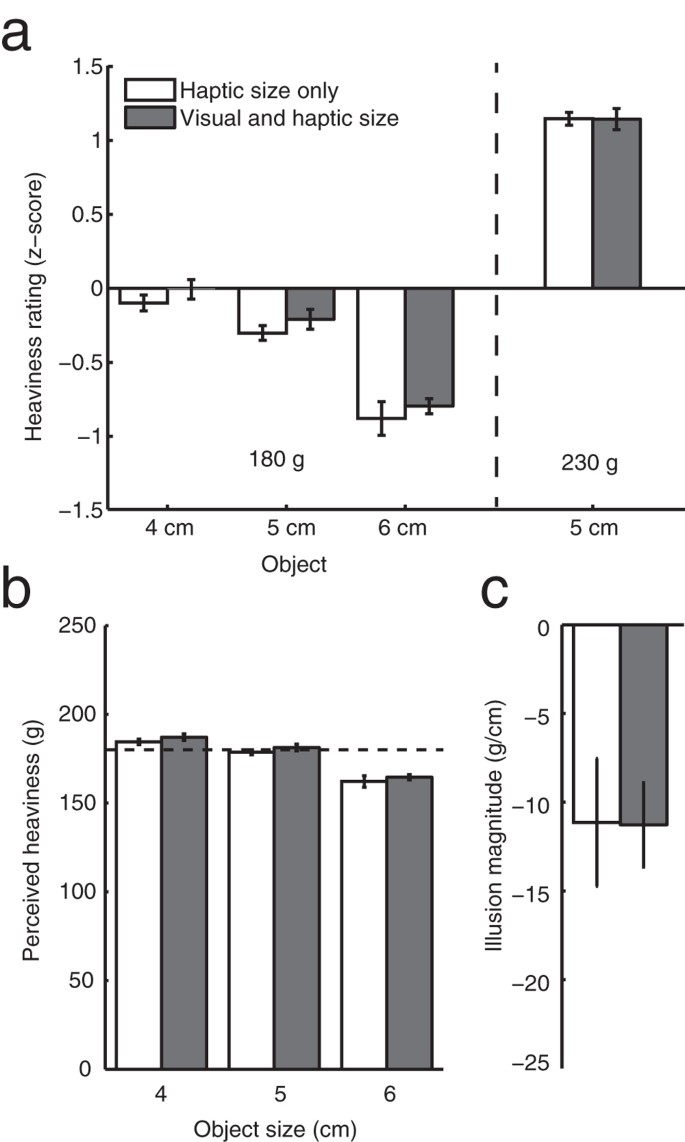



Object Size Can Influence Perceived Weight Independent Of Visual Estimates Of The Volume Of Material Scientific Reports
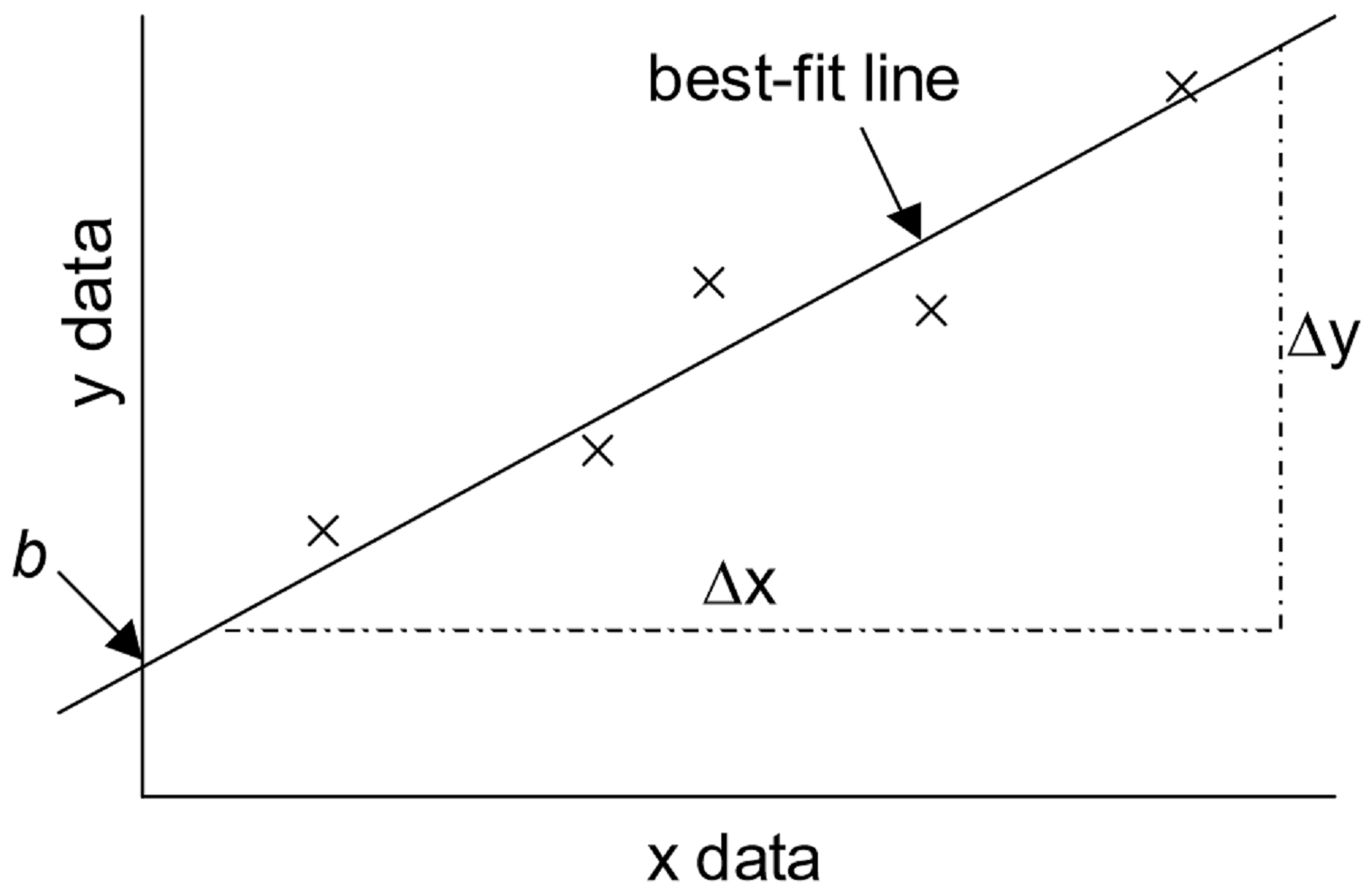



1 Using Excel For Graphical Analysis Of Data Experiment Chemistry Libretexts
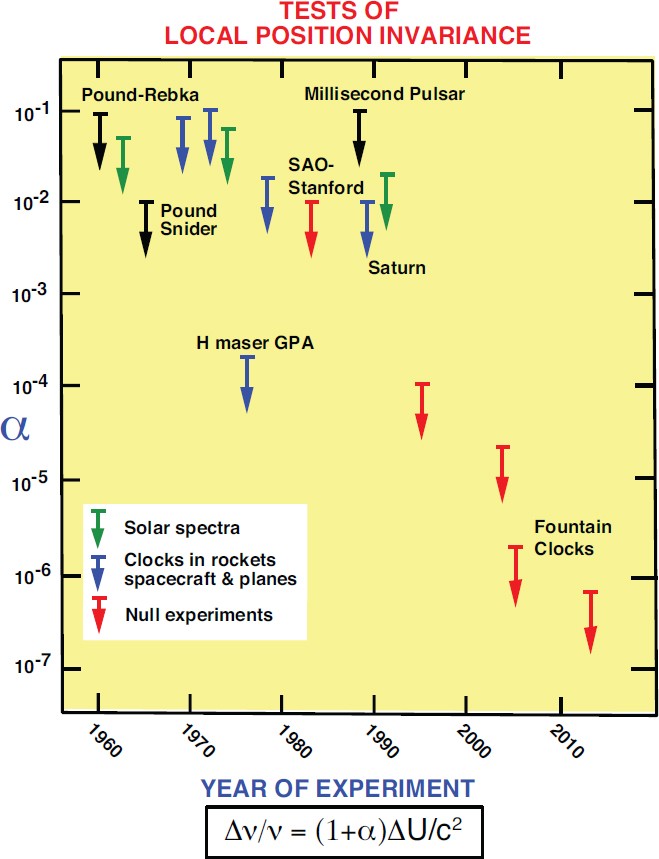



The Confrontation Between General Relativity And Experiment Springerlink




How Do Scientists Measure Or Calculate The Weight Of A Planet Scientific American




Temperature Change And Heat Capacity Physics
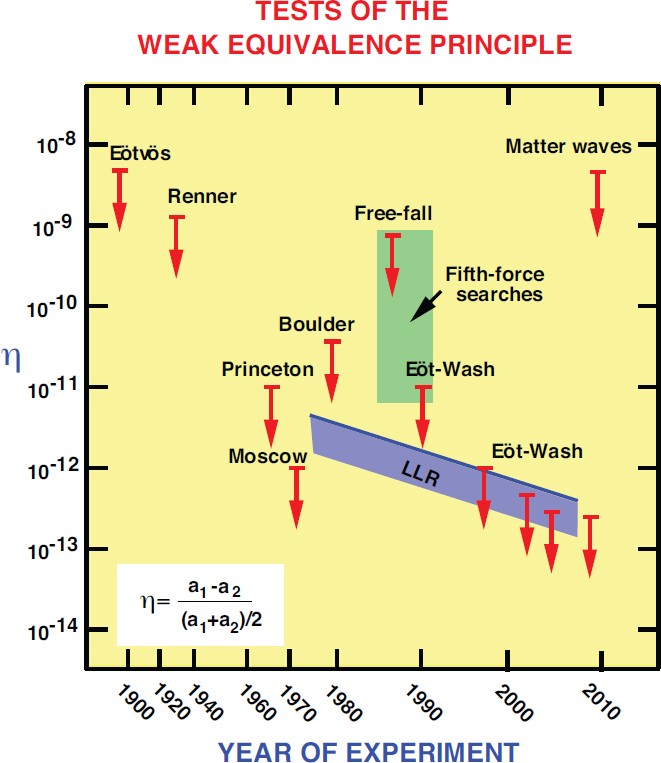



The Confrontation Between General Relativity And Experiment Springerlink




Experimental Design An Overview Sciencedirect Topics




A Mass Density Model Can Account For The Size Weight Illusion




Scientific Method Wikipedia




The Material Weight Illusion Disappears Or Inverts In Objects Made Of Two Materials Journal Of Neurophysiology
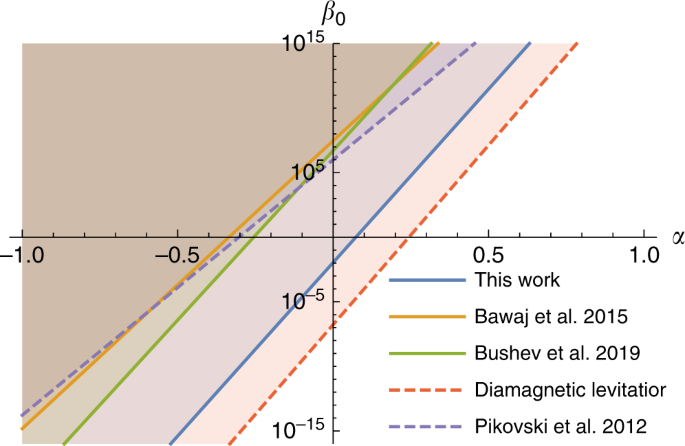



On Quantum Gravity Tests With Composite Particles Nature Communications



0 件のコメント:
コメントを投稿